Pisum Genetics
2007—Volume 39
Brief Communications
Pea mutant line sprint-2Nod-3 represents
a new mutant allele of pea symbiotic gene sym19
a new mutant allele of pea symbiotic gene sym19
Zhukov V.A., Borisov A.Y., Tikhonovich I.A. Lab. of Gen. of Plant-Microbe Interactions
(http://www.arriam.spb.ru/eng/lab9) All-Russia Res. Inst. for Agri. Microbiol., St. Petersburg, Russia
Pea (Pisum sativum L.) remains an important model object
studying genetics of symbiotic systems, despite the fact that it
a large genome and a relatively low capability for genetic
transformation. A large collection of well-characterized
symbiotic mutants with unique phenotypic manifestation has
been obtained (1).
studying genetics of symbiotic systems, despite the fact that it
a large genome and a relatively low capability for genetic
transformation. A large collection of well-characterized
symbiotic mutants with unique phenotypic manifestation has
been obtained (1).
Here we describe the mutant Sprint-2Nod-- 3 obtained in the
background of laboratory line Sprint-2 after EMS mutagenesis
background of laboratory line Sprint-2 after EMS mutagenesis
for
has
has
more than 15 years ago. The line Sprint-2Nod--3 is derived from
1
M2 plant that had gray and green nodules and was deficient in
nitrogen fixation (Fix- phenotype). Four plants of the next
generation (M3) also had gray and green nodules, but all the
nitrogen fixation (Fix- phenotype). Four plants of the next
generation (M3) also had gray and green nodules, but all the
progenies in M4 totally lacked nodules. The line was finally
phenotypically characterized as Nod- mutant forming specific
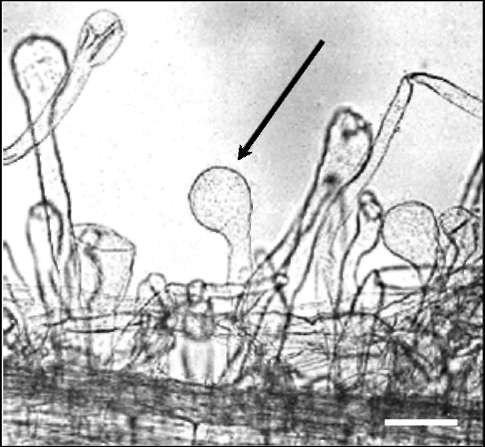
deformations of root hairs resembling drumsticks by shape (Fig.
phenotypically characterized as Nod- mutant forming specific
deformations of root hairs resembling drumsticks by shape (Fig.
Fig. 1. A characteristic deformation of
root hair tips in Sprint-2Nod-3 mutant
(sym19). Arrow points to deformed root
hair tip. Scale bar 0.05 mm
root hair tips in Sprint-2Nod-3 mutant
(sym19). Arrow points to deformed root
hair tip. Scale bar 0.05 mm
1).
This phenotype is specific for pea mutants in sym8 and sym19
genes (2, 3). Allelism tests with pea lines carrying mutations in "early" symbiotic genes sym8, sym9, sym10 and
sym19 demonstrated that the line Sprint-2Nod--3 is a mutant of sym19.
genes (2, 3). Allelism tests with pea lines carrying mutations in "early" symbiotic genes sym8, sym9, sym10 and
sym19 demonstrated that the line Sprint-2Nod--3 is a mutant of sym19.
How could a Fix-mutant turn out to be a Nod--phenotype? The explanation we could propose is the
following. The initial M2 plant (Fix-) was grown in summer under "outdoor" conditions, where the temperature
varied from 10°C to 25°C. Next winter M3 plants (Fix-) were grown in a greenhouse where the temperature also
varied from 10°C to 25°C. All the following generations were cultivated in the climatic chamber (Vb'tsch
Industrietechnik VB 1014, Germany) under strictly controlled temperature conditions (21 ± 1°C) and
demonstrated Nod--phenotype. Lowering the night temperature to 15°C is usually enough for manifestation of
temperature-sensitive phenotype in pea symbiotic mutants. Therefore, we suggest that Sprint-2Nod-- 3 is an
example of temperature-sensitive mutation of sym19.
following. The initial M2 plant (Fix-) was grown in summer under "outdoor" conditions, where the temperature
varied from 10°C to 25°C. Next winter M3 plants (Fix-) were grown in a greenhouse where the temperature also
varied from 10°C to 25°C. All the following generations were cultivated in the climatic chamber (Vb'tsch
Industrietechnik VB 1014, Germany) under strictly controlled temperature conditions (21 ± 1°C) and
demonstrated Nod--phenotype. Lowering the night temperature to 15°C is usually enough for manifestation of
temperature-sensitive phenotype in pea symbiotic mutants. Therefore, we suggest that Sprint-2Nod-- 3 is an
example of temperature-sensitive mutation of sym19.
27