Combination of mutations sil and ins2 can cause
conversion of stipules into compound
leaves
Berdnikov,
V.A. and Gorel, F.L.
Inst. of Cytology and Genetics
Russian Acad. of Sci., Novosibirsk,
Russia
Previously
we described the ins2 mutation, which
caused angular incisions in the leaflet tips (1). Within such incisions, the
central veins of the leaflets were transformed into rachillae bearing unbranched
tendrils. This phenotype becomes more pronounced from node to node, up the
plant. At flowering nodes, leaflets may lose their laminae and be converted into
compound pinnae typical for af plants.
In the ins2/ins2 tl/tl double
homozygote, the structure formed at the base of leaflet incision may look like
the pinnate leaf of a tl mutant,
specifically a rachis bearing normal oval leaflets.
 It was shown
by Marx (3) that, in plants homozygous for the mutation sinuate
leaves (sil), the distal portions of stipules acquire some properties of
leaflets. In plants homozygous for sil,
the chimeric nature of stipules can be easily detected in the background of
mutations differentially affecting the wax coat of stipules and leaflets. For
example, the wlo (supra-incerata)
mutation, removes wax from the upper surface of leaflets and also from the
leaflet-like distal portion of the sil
stipules (3). Most impressively the sil
effects are manifested in the af/af
background (3): the stipules of sil/sil af/af
plants are often split with adventitious structures looking like an af
leaf arising from the base of the cleft. Recently, Husbands et al. (2) described
effects of sil on the stipule phenotypes in Af/Af and af/Af backgrounds.
They showed that under short day conditions the stipules of sil/sil,
af/Af plants were converted into compound leaves with their own stipules. In
present paper we describe a similar transformation of the stipules in plants
homozygous for sil and ins2.
It was shown
by Marx (3) that, in plants homozygous for the mutation sinuate
leaves (sil), the distal portions of stipules acquire some properties of
leaflets. In plants homozygous for sil,
the chimeric nature of stipules can be easily detected in the background of
mutations differentially affecting the wax coat of stipules and leaflets. For
example, the wlo (supra-incerata)
mutation, removes wax from the upper surface of leaflets and also from the
leaflet-like distal portion of the sil
stipules (3). Most impressively the sil
effects are manifested in the af/af
background (3): the stipules of sil/sil af/af
plants are often split with adventitious structures looking like an af
leaf arising from the base of the cleft. Recently, Husbands et al. (2) described
effects of sil on the stipule phenotypes in Af/Af and af/Af backgrounds.
They showed that under short day conditions the stipules of sil/sil,
af/Af plants were converted into compound leaves with their own stipules. In
present paper we describe a similar transformation of the stipules in plants
homozygous for sil and ins2.
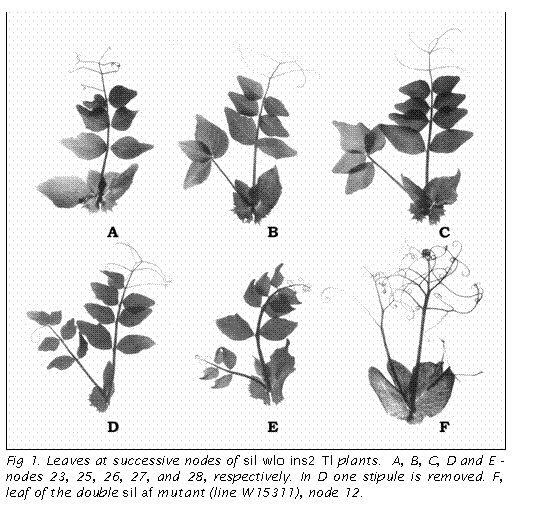
In
the double sil af mutants (lines
W15110 and W15311 from the Marx collection at the USDA Western Regional Plant
Introduction Station, Pullman, Washington), we observed that the leaflet-like
segment of the stipule is split and its main vein is transformed into a
structure indistinguishable from the distal part of a compound pinna typical for
the af leaf (Fig. 1F). At the same
time, in contrast to observation of Husbands et al (2), in sil
wlo Af plants (line W15460 from the Marx collection) we never found cleaved
stipules, although the stipules always had the waxless diamond-shaped regions at
their tips. Subsequently, we will
refer to these regions covered with cuticle typical for leaflets, as leaflet-like
regions (LLR).
Plants
were grown in the greenhouse in hydroponic claydite beds fed by standard Knop
nutrient solution under long day conditions. They were illuminated by 8 hr
daylight/ 16 hr incandescent light of 7,000 - 10,000 lx intensity. One plant of
the line W15460 (sil wlo tl Ins2) was
crossed with our line AFD (Sil Wlo Tl ins2),
and among 152 F2 plants six individuals with incisions on leaflets
and waxless LLRs on the stipules were selected. All of them had tendrils with
narrow laminae characteristic for the heterozygotes tl/Tl. Among their
progenies from selfing several exceptional individuals with a strikingly altered
stipule morphology were observed, although their leaflets were practically
devoid of incisions. Two such plants (one homozygous for tl
and one homozygous for Tl) were
selected for further analysis. All the offspring from the cross of exceptional
plants with the lines bearing wild-type Ins2
alleles had cuts in the leaflets that indicated that the sil
parents were homozygous for ins2.
 Fig. 1 shows
the changes in stipule morphology in the triple homozygote ins2/ins2
sil/sil wlo/wlo. At higher nodes (>15) the stipule is often transformed
into a tripartite structure, two lateral stipule-like parts and the central one
developed from the waxless LLR. The central part tends to be more complex at
successive nodes. Several stages of complexity can be distinguished. First, the
waxless LLR separates as a lobe which looks like a large leaflet with a short
petiolule resembling the leaf of the uni
mutant (Fig. 1A); 2) LLR is converted into a compound leaf without tendrils
(Fig. 1B,C); 3) LLR becomes similar to the leaf of unitac
mutant with the terminal leaflet and subterminal tendrils (Fig. 1D); and 4) LLR
is transformed into an adventitious compound wild-type leaf with the terminal
tendril (Fig. 1E). Thus, we can see that the extreme form of stipule
transformation looks like the wild-type leaf with both stipules. The maximum
expression of aberrant phenotype is observed at pre-flowering nodes. It should
be noted, that the leaves subtending flowers have normal stipules.
Fig. 1 shows
the changes in stipule morphology in the triple homozygote ins2/ins2
sil/sil wlo/wlo. At higher nodes (>15) the stipule is often transformed
into a tripartite structure, two lateral stipule-like parts and the central one
developed from the waxless LLR. The central part tends to be more complex at
successive nodes. Several stages of complexity can be distinguished. First, the
waxless LLR separates as a lobe which looks like a large leaflet with a short
petiolule resembling the leaf of the uni
mutant (Fig. 1A); 2) LLR is converted into a compound leaf without tendrils
(Fig. 1B,C); 3) LLR becomes similar to the leaf of unitac
mutant with the terminal leaflet and subterminal tendrils (Fig. 1D); and 4) LLR
is transformed into an adventitious compound wild-type leaf with the terminal
tendril (Fig. 1E). Thus, we can see that the extreme form of stipule
transformation looks like the wild-type leaf with both stipules. The maximum
expression of aberrant phenotype is observed at pre-flowering nodes. It should
be noted, that the leaves subtending flowers have normal stipules.
In
the sil ins2 tl wlo mutant, the adventitious leaf developed from LLR looked like
a unipinnate leaf of the tl mutant
(Fig. 2). It is of interest to note that the leaflets as a rule lacked incisions
in spite of the presence of ins2. The
first flowers appear very late (at nodes > 30), and the leaves subtending
flowers are normal. Often the plants do not flower at all.
Earlier
we showed that Ins2 is a synergist of Af,
and its effect becomes stronger in the af/Af
background (1). Stipule phenotypes of the sil
ins2 plants (Fig. 1) display a striking resemblance to those of the sil/sil af/Af plants presented by Husbands et al (2). It seems that
the homozygote for ins2 can mimic the
action of the heterozygote af/Af.
Thus, using combination of sil
and ins2 mutations, we showed that the original cluster of three stem
appendages (one rachis and two stipules) tend to transform into seven
appendages, three rachises and four stipules. It is important to notice that the
stipules between rachises have a leaflet-like sector and in principle may
generate new rachises and stipules between them. Thus, in sil mutants, there is a morphogenetic potency to initiate
repetitively primordia of rachises and stipules around the shoot apex. The
continuation of this morphogenetic activity may eventually result in formation
of a whorl of alternating leaves and stipules. The generation of such whorls
during ontogeny of Acacia longipedunculata
was described by Rutishauser and Sattler (4). Thus, orthologs of Sil
may have some relation to genetic mechanisms of nodal whorl formation in
evolution.
Acknowledgment:
This work was supported by the Russian State Program
'Russian Fund for Fundamental Research', grant No 02-04-49426.
1. Berdnikov,
V.A., Gorel’, F.L., Bogdanova, V.S. and Kosterin, O.E. 2000. Pisum Genetics
32: 9-12.
2. Husbands,
A., Emirzade, T. and DeMason, D. 2003.
Pisum Genetics 35: 6-9.
3. Marx,
G.A. 1977.
Amer. J. Bot. 64: 273-277.
4. Rutishauser,
R. and Sattler, R. 1986. Can. J. Bot. 64: 1987-2019.
 It was shown
by Marx (3) that, in plants homozygous for the mutation sinuate
leaves (sil), the distal portions of stipules acquire some properties of
leaflets. In plants homozygous for sil,
the chimeric nature of stipules can be easily detected in the background of
mutations differentially affecting the wax coat of stipules and leaflets. For
example, the wlo (supra-incerata)
mutation, removes wax from the upper surface of leaflets and also from the
leaflet-like distal portion of the sil
stipules (3). Most impressively the sil
effects are manifested in the af/af
background (3): the stipules of sil/sil af/af
plants are often split with adventitious structures looking like an af
leaf arising from the base of the cleft. Recently, Husbands et al. (2) described
effects of sil on the stipule phenotypes in Af/Af and af/Af backgrounds.
They showed that under short day conditions the stipules of sil/sil,
af/Af plants were converted into compound leaves with their own stipules. In
present paper we describe a similar transformation of the stipules in plants
homozygous for sil and ins2.
It was shown
by Marx (3) that, in plants homozygous for the mutation sinuate
leaves (sil), the distal portions of stipules acquire some properties of
leaflets. In plants homozygous for sil,
the chimeric nature of stipules can be easily detected in the background of
mutations differentially affecting the wax coat of stipules and leaflets. For
example, the wlo (supra-incerata)
mutation, removes wax from the upper surface of leaflets and also from the
leaflet-like distal portion of the sil
stipules (3). Most impressively the sil
effects are manifested in the af/af
background (3): the stipules of sil/sil af/af
plants are often split with adventitious structures looking like an af
leaf arising from the base of the cleft. Recently, Husbands et al. (2) described
effects of sil on the stipule phenotypes in Af/Af and af/Af backgrounds.
They showed that under short day conditions the stipules of sil/sil,
af/Af plants were converted into compound leaves with their own stipules. In
present paper we describe a similar transformation of the stipules in plants
homozygous for sil and ins2.
