 Introduction
Introduction
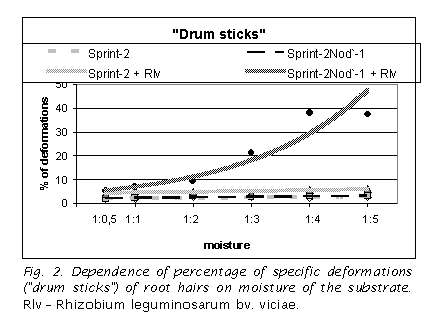
“Drum sticks” is a trait associated with the Sym8 locus in pea
 Introduction
Introduction
The gene sym8 controls the earliest developmental stages of pea symbioses with nodule bacteria and endomycorrhizal fungi (2, 4, 5). Mutants in this gene do not form characteristic root hair curling upon interactions with nodule bacteria and abort colonizing hyphae growth of endomycorrhizal fungi just after appressoria formation. To date, 11 independently obtained mutant alleles of the gene sym8 in three different genetic backgrounds have been identified using experimental mutagenesis (3). A characteristic trait of the mutants in this gene is a formation of specific spheric swelling of the root hair tip, in the way the root hairs resemble “drum sticks” by shape (4) (Fig. 1). It was assumed that specific deformations of root hair tips depend on the moisture of the substrate (4). To check this hypothesis, the phenotypic manifestation of this characteristic trait under different moisture conditions and in absence or in presence of rhizobia was analyzed.
Materials and methods
Seeds of pea lines Sprint-2 Nod--1 (sym8) and Sprint-2 (wild type) were sterilized by concentrated sulfuric acid, washed by sterile water, and soaked overnight in water aerated by bubbling and then planted into Petri dishes with sterile vermiculite. Different quantities of distilled autoclaved water were added to reach different moisture level of the substrate. Six variants of substrate moisture (vermiculite/water ratio 1:0.5, 1:1, 1:2, 1:3, 1:4, 1:5 w/v) were examined. Five seeds of each variant were cultivated in a thermostat at 28oC. Seeds were inoculated with Rhizobium leguminosarum bv. viciae, strain CIAM1026, 1 ml of inoculum (108-109 cells) per seed. The analysis was performed at 3 and 6 days after planting (DAP).
The main roots of seedlings were stained with 0.01% methylene blue and then analyzed using light microscope Opton Axiovert-35. The relative quantity of deformed root hairs was estimated and evaluated in percents. At least 20 fields of vision per root were analyzed.
Results and discussion
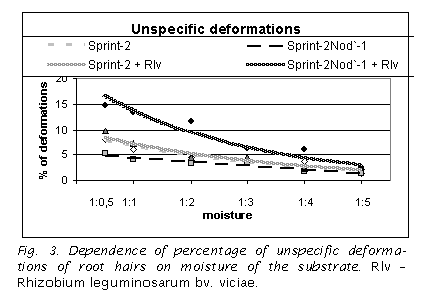
It was shown that under sterile conditions the mutant does not differ from the wild type line at both 3 (data not shown) and 6 DAP (Fig. 2, 3). The number of specific deformations (drum sticks) was very low and it was independent on the level of the substrate moisture (Fig. 2). The number of unspecific deformations of different shape was decreased with the increase in the moisture level (Fig. 3).
In presence of nodule bacteria the striking difference between mutant and wild type line was observed. The number of drum sticks in the mutant, but not in the wild type line, swiftly increased in parallel with the increase in moisture content of the substrate (Fig. 2). In wild type the inoculation did not change the number of specific and unspecific deformations in comparison with non-inoculated control (Fig. 2, 3).
Thus, it was shown that the mutant phenotype becomes pronounced only in presence of nodule bacteria, and its manifestation depends on the substrate moisture. That means that mutation in the gene sym8 changes the root hair tip membrane permeability upon perception of the Nod-factor signal of nodule bacteria. This result is intriguing because sym8 is suggested to be an ortholog of DMI1 of Medicago truncatula Gaertn., which codes for a component of the ligand gated cation channel (1).
 Acknowledgements:
This
work was supported by RAAS and grants of RFBR (04-04-48457-а),
President of Russia (MD-350.2003.04, HIII-1103.2003.04), and
CRDF (ST-012-0).
Acknowledgements:
This
work was supported by RAAS and grants of RFBR (04-04-48457-а),
President of Russia (MD-350.2003.04, HIII-1103.2003.04), and
CRDF (ST-012-0).
1. Ané, J.M., Kiss, G.B., Riely, B.K , Penmetsa, R.V., Oldroyd, G.E.D., Avax, C., Levy, J., Debellé, F., Baek, J.-M., Kalo, P., Rosenberg, C., Roe, B.A., Long, S.R., Dénarié, J. and Cook, D.R. 2004. Science 303: 1364-1367.
2. Balaji, B., Ba, A.M., LaRue, T.A., Tepfer, D. and Piche, Y. 1994. Plant Sci. 102: 195-203.
3. Borisov, A.Y., Barmicheva, E.M., Jacobi, L.M., Tsyganov, V.E., Voroshilova, V.A. and Tikhonovich, I.A. 2000. Czech J. Genet. Plant Breed. 36: 106-110.
4. Borisov, A.Y., Rozov, S.M., Tsyganov, V.E., Kulikova, O.A., Kolycheva, A.N., Yakobi, L.M., Ovtsyna, A.O. and Tikhonovich, I.A. 1994. Russ. J. Genet. 30: 1284-1292.
5. Kolycheva, A.N., Jacobi, L.M., Borisov, A.Y., Filatov, A.A., Tikhonovich, I.A. and Muromtsev, G.S. 1993. Pisum Genetics 25: 22.