The
aero2 (aeromaculata2) mutation in pea
increases
leaf flecking and complexity but,
unlike aero1, does not promote
flowering
Murfet,
I.C. and
Pte. Bag 55, Hobart, TAS 7001, Australia
Taylor,
S.A.
School
of Plant Sci.
Univ. of Tasmania, Hobart, Australia
The aero1 (aeromaculata1) mutation results in more extensive expression of the
patches of silver flecking (aeromaculata) normally seen on pea leaves leading to
a phenotype known as supaeromaculata (2, 5). While aero1 mutants are characterized by increased leaf flecking, they
also display a strong pleiotropic phenotype that includes shorter internodes,
reduced stature and lower yield (10, 12). In addition, flowering in aero2
plants is strongly promoted in terms of both node of flower initiation and time
to first open flower; fewer leaves are produced before the onset of apical
arrest and senescence; and the increases in leaf complexity that occur during
ontogeny occur at lower nodes than in wild-type (WT) plants (12).
The heteroblastic increase in leaf complexity in pea has been examined
extensively (4, 13, 14). Scale leaves occur at nodes 1 and 2. The first true
leaf occurs at node 3 and consists of a pair of basal stipules and a rachis
bearing a pair of opposite leaflets and a simple terminal tendril. Above node 3,
leaf complexity increases with the addition of pairs of tendrils and leaflets.
The leaves of adult WT plants possess two to three pairs of proximal leaflets,
two to three pairs of distal simple tendrils, and a terminal simple tendril
(13). The node at which the first leaf with 4 leaflets (C-4) occurs may be some
30-40 % lower in aero1 mutants than in
their WT isolines (12).
We have classified aero1 as a
heterochronic mutant and have argued that the broad spectrum of pleiotropic
traits may all reflect the acceleration, relative to the WT, of various
developmental steps during the course of ontogeny (12). Acceleration is obvious
in the case of traits like earlier flowering, senescence and leaf change but
less obvious in the case of increased leaf flecking. However, the silver flecks
are underlain by sub-epidermal air spaces (11), and the more extensive flecking
of aero1 leaves may arise through an
acceleration of certain anatomical or biochemical changes during development of
the leaf (12).
Supaeromaculata mutants are not uncommon and at least ten aero1
alleles are known (2, 10, 12). In this paper, we report on a new recessive
supaeromaculata mutant Dr J.L. Weller obtained at Hobart
in an M2 population following EMS
treatment of cv. Torsdag (HL107). We cleaned up the new mutant by several
generations of single-plant selection and two generations of backcrossing to cv.
Torsdag, before including it in the Hobart pea collection as HL303. F2 and F3 data from the two
backcrosses confirmed single-gene recessive inheritance. The combined F2
data of 80 WT and 34 mutant plants are in accordance with a 3:1 ratio (P >
0.2) and mutant F2 plants bred true in F3. There was
evidence the WT allele was not fully dominant over the mutant allele. Visual
separation of WT F2 plants into two classes based on normal versus
slightly elevated flecking intensity approximated a 1:2 ratio. However, there
was no clear difference between the two groups and a few doubtful plants were
arbitrarily assigned to one or other class. In contrast, mutant F2
plants were visually distinct.
We made reciprocal crosses between the new aero
mutant and mutants aero1-1 and aero1-10
. Six F1 plants were grown from each cross. All 12 F1
plants had a WT phenotype. We conclude the new mutant is not allelic with aero1
and have named the new gene AERO2 (AEROMACULATA2)
with HL303 as the type line for mutant allele aero2-1.
The supaeromaculata phenotype of aero2-1
is slightly weaker than that of aero1-10,
and much weaker than the aero1-1
phenotype (Fig. 1). In young seedlings, the increased flecking of aero1-1
plants was clearly evident at day 10 from
the stipules and leaflets of the first foliage leaf (node 3). In contrast, the
increased flecking of aero1-10
plants was not fully evident until expansion of the stipules and leaflets at
node 5 at around two weeks from sowing. For aero2-1 plants, some increase in flecking could be seen on the
stipules at node 5 but an obvious supaeromaculata phenotype was not fully
apparent until expansion of the leaves at nodes 6 and 7.
 Following recognition of the interesting and very strong pleiotropic
phenotype of aero1 (10, 12), we made a
detailed phenotypic comparison between WT and aero2
segregates in the F2 of the first and second backcrosses between the
mutant and cv. Torsdag. Both backcrosses gave consistent results. All plants
were grown in the glasshouse at Hobart in individual 14-cm slimline pots under an 18-h photoperiod. Further details on
growing conditions are as described earlier (12).
Following recognition of the interesting and very strong pleiotropic
phenotype of aero1 (10, 12), we made a
detailed phenotypic comparison between WT and aero2
segregates in the F2 of the first and second backcrosses between the
mutant and cv. Torsdag. Both backcrosses gave consistent results. All plants
were grown in the glasshouse at Hobart in individual 14-cm slimline pots under an 18-h photoperiod. Further details on
growing conditions are as described earlier (12).
The aero2 mutant also displayed
a wide range of pleiotropic traits but with some major differences from the
traits of aero1 mutants.
The aero2 mutation certainly
accelerated increases in leaf complexity. The transition to 4- and 6-leaflet
leaves occurred around 12% earlier in aero2
than WT plants (P < 0.001, Table 1). All aero2
plants attained
6-leaflet
leaves and 38% went on to produce leaves with a supranormal state of 7 or 8
leaflets (Table 2).
In
contrast,
only 90% of WT plants produced leaves with 6 leaflets, and not one produced
leaves with 7 or 8 leaflets (Table 2).
In contrast to the aero1
mutant, flowering was not promoted in aero2
plants. The node of flower initiation was slightly higher (4% increase) and
number of days to first open flower slightly increased (6%) in aero2
plants compared with WT plants; both differences are significant (P < 0.001,
Table 1). The rate of leaf appearance was 2% less (P < 0.01) in aero2
plants contributing to the increase in flowering time. While these 2-6%
differences are numerically small, they were statistically significant and very
similar results were obtained in the second backcross (data not shown). Thus we
believe they are a true effect of the aero2
mutation.
Stem length between nodes 1 and 4 and 1 and 9 was around 25% less in aero2
than WT plants (Table 1). The effect of the aero2
mutation on internode length continued throughout the life of the plant.
The average internode length of mature plants (excluding second growth)
was around 20% less in aero2 than WT plants in both backcross F2 populations,
and individual plots of this trait for aero2
and WT plants did not overlap in either F2. The anatomical basis for
the 28% reduction in the length of internode 8 (between nodes 8 and 9) was
examined using the epidermal strip technique of Arney and Mancinelli (1). The
data in Table 1 are based on measurement of 10 epidermal cells per plant from 6
plants of each phenotype. They show an 18% reduction in cell number (P <
0.01) and an 11% reduction in cell length (P > 0.05) in aero2 plants. These results indicate reduced cell division as the
major cause of the reduced internode length, although reduced cell elongation
seems also to have contributed. The width of internode 8 was similar in WT and aero2
plants (Table 1).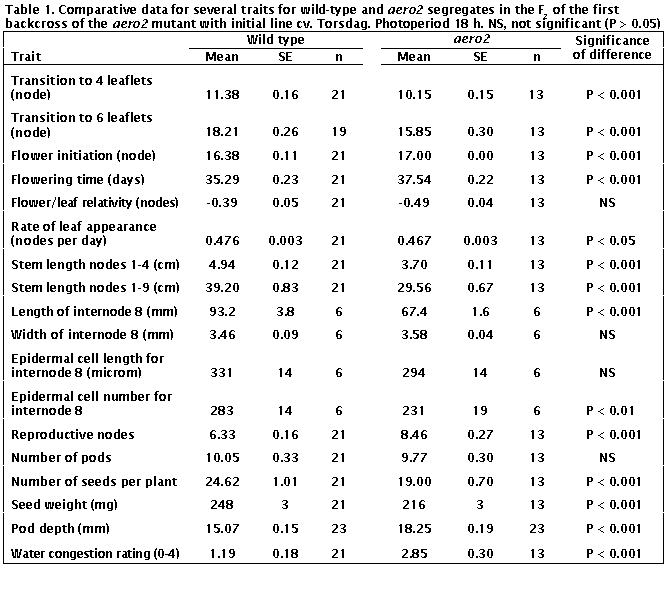
Although the aero2 plants produced two more reproductive nodes than WT plants in
the first reproductive cycle (P < 0.001), both genotypes produced a similar
number of pods (P > 0.5) (Table 1). The majority of WT  plants (81%, 17/21) underwent normal
monocarpic senescence at the end of the first reproductive cycle. However, the
majority of aero2 plants (92%, 12/13),
after slowing to near zero growth, recommenced active growth and entered a
second reproductive cycle. This difference is significant at P < 0.001. The
failure of the aero2 plants to undergo
whole-plant senescence at the end of the first reproductive cycle may have
resulted from their reduced reproductive load. While the aero2 plants had the same number of pods as WT plants, they had 23%
fewer seeds (P < 0.001) and the seeds weighed on average 13% less than seeds
from WT plants (P
< 0.001). The relationship between reduced reproductive load and failure to
undergo whole-plant senescence after a single reproductive cycle was examined
earlier in pea in respect of the ar
and n mutants (7).
plants (81%, 17/21) underwent normal
monocarpic senescence at the end of the first reproductive cycle. However, the
majority of aero2 plants (92%, 12/13),
after slowing to near zero growth, recommenced active growth and entered a
second reproductive cycle. This difference is significant at P < 0.001. The
failure of the aero2 plants to undergo
whole-plant senescence at the end of the first reproductive cycle may have
resulted from their reduced reproductive load. While the aero2 plants had the same number of pods as WT plants, they had 23%
fewer seeds (P < 0.001) and the seeds weighed on average 13% less than seeds
from WT plants (P
< 0.001). The relationship between reduced reproductive load and failure to
undergo whole-plant senescence after a single reproductive cycle was examined
earlier in pea in respect of the ar
and n mutants (7).
The depth of aero2 pods
exceeded that of WT pods by around 20 % in the first backcross F2
(Table 1) and 10 % in the second backcross F2 (data not shown). The aero2
pods tended to be shorter than WT pods but pod length would certainly be
influenced by the fact that aero2 pods
contained on average 20 % fewer seeds than the WT pods (Table 1).
A period of damp overcast weather during the growth of the first
backcross F2 resulted in some water congestion damage (see 8) to the
plants. Water congestion damage was assessed on a subjective scale of 0 (no
damage) to 4 (expansion of leaflet lamina fully suppressed). The aero2 plants proved to be much more susceptible to water congestion
than the WT plants (Table 1). For aero2
plants the worst affected leaf was estimated, on average, to have lost about 75%
of its potential lamina area compared with around 25% for WT plants. Any
difference in susceptibility to water congestion was not exposed in the second
backcross F2 as all plants were free of damage.
 In addition to the effects on leaf flecking, leaf complexity, and leaf
susceptibility to water congestion damage, the aero2 mutation may also have caused some unusual morphological
features in one or more leaves starting at or just above the node of flower
initiation. In the second backcross F2, all four aero2
plants had one or more leaflets where the leaflet base was rolled up into a very
short conical funnel (Fig. 2), and two of the four aero2
plants had one leaf where a leaflet appeared to be replaced by a short (around 5
mm), slender cylinder of translucent tissue, which, for want of a better term,
we have referred to as a pin (Fig. 3). The funnel trait expressed in one or both
of the first pair of leaflets. The pin trait expressed opposite a normal leaflet
at the third pinna-pair position numbering from the proximal end of the rachis.
These unusual leaf features were not observed in any of the 16 WT siblings.
Unfortunately, we did not scan the much larger first backcross F2 for
these unusual features, so the observation rests on only four aero2
F2 plants. However, weak funnels have subsequently been observed in
HL303 plants (HL303 is descended from one of these four aero2
plants).
In addition to the effects on leaf flecking, leaf complexity, and leaf
susceptibility to water congestion damage, the aero2 mutation may also have caused some unusual morphological
features in one or more leaves starting at or just above the node of flower
initiation. In the second backcross F2, all four aero2
plants had one or more leaflets where the leaflet base was rolled up into a very
short conical funnel (Fig. 2), and two of the four aero2
plants had one leaf where a leaflet appeared to be replaced by a short (around 5
mm), slender cylinder of translucent tissue, which, for want of a better term,
we have referred to as a pin (Fig. 3). The funnel trait expressed in one or both
of the first pair of leaflets. The pin trait expressed opposite a normal leaflet
at the third pinna-pair position numbering from the proximal end of the rachis.
These unusual leaf features were not observed in any of the 16 WT siblings.
Unfortunately, we did not scan the much larger first backcross F2 for
these unusual features, so the observation rests on only four aero2
F2 plants. However, weak funnels have subsequently been observed in
HL303 plants (HL303 is descended from one of these four aero2
plants).
The funnel and pin features give the impression that leaflet and lamina
development has been partially or wholly truncated in these cases, which fits
well with the idea that the aero2
mutation is hastening the timing of various events during development of
vegetative organs.
Funnel leaflets and pins/needles are the defining features of the lld
(leaflet development) pea mutant (9). The strongest expression of the
funnel-leaflet trait in aero2 plants
(Fig. 2) was approximately equal to the weakest expression of the funnel-leaflet
trait illustrated for lld plants (Fig.
1B in 9). Interestingly, the peak expression of the lld
phenotype occurred as the plants entered the reproductive phase (9), and
expression of funnels and/or pins in aero2
plants commenced at or just above the node of flower initiation. Again, the
strongest expression of the lld
phenotype occurred at position 3 along the rachis (9) and that is where the pins
(complete lamina suppression?) occurred in aero2
plants (Fig. 3). Thus the leaf phenotypes of lld
and aero2 plants share certain
similarities, both in morphological features expressed (funnels, pins) and in
the timing and place of maximum expression. However, LLD
has a more confined role in leaf development than AERO2 as lld plants do not
appear to have increased leaf flecking.
 The adt (air dots) pea mutant has a phenotype that includes numerous tiny
grey spots along the veins of leaflets and stipules, shorter and wider pods, and
the terminal tendril of a leaf is often replaced by a leaflet (3). Thus the aero2
and adt mutations both increase leaf flecking and alter pod dimensions
but overall the two mutants are quite distinct.
The adt (air dots) pea mutant has a phenotype that includes numerous tiny
grey spots along the veins of leaflets and stipules, shorter and wider pods, and
the terminal tendril of a leaf is often replaced by a leaflet (3). Thus the aero2
and adt mutations both increase leaf flecking and alter pod dimensions
but overall the two mutants are quite distinct.
Comparison of the list of phenotypic effects of the aero1
and aero2 mutations reveals a number
of features in common. Both mutations appear to accelerate changes that affect
vegetative traits like internode length and leaf complexity. The anatomical data
(Table 1) show that reduced cell division is the major cause of the shorter
internodes in the aero2 mutant. This
could indicate that cell division ceases earlier in the development of the aero2
internodes than in the WT internodes.
The accelerated transition to 4- and 6-leaflet leaves certainly
categorizes aero2 as a heterochronic
mutant as the timing of these events is brought forward relative to the
situation in the WT ancestor. The effect of the aero2
mutation on leaf complexity is further emphasized by the occurrence of
supranormal 8-leaflet leaves on some aero2
plants. WT plants (with normal leaf flecking) do sometimes attain 8-leaflet
leaves when grown under short-day conditions that allow vigorous growth and a
prolonged vegetative phase but we have never observed 8-leaflet leaves on WT
plants grown under the 18-h conditions used in this study.
The accelerated transition to 4- and 6-leaflet leaves was only about
one-third as strong in aero2-1 (Table
1) as in the aero1-1 and aero1-10 mutants (12). However, the aero1 data were based on comparison of homozygous WT and mutant
isolines, where as the WT plants in the aero2
study were comprised of a mixture of homozygous and heterozygous plants. Any
degree of partial dominance would diminish the observed difference. The effect
on leaf flecking was also weaker in aero2-1
than in the aero1 mutants (Fig. 1).
Our ability to comment further on the weaker action of aero2-1 is limited by the fact that only one aero2 mutant is currently available and we do not know whether aero2-1
is a null or leaky allele.
The aero1 mutation
significantly promotes flowering as shown by several measures: there is a major
reduction in the node of flower initiation and the time to first open flower;
the size of the quantitative response to photoperiod is substantially diminished
in aero1 plants compared with WT
plants; and development of the flower bud is accelerated relative to leaf
development as evidenced by the higher flower/leaf relativity values (see 6) in aero1
plants (12). In contrast, the aero2
mutation does not promote flowering by any measure. On the contrary, we found a
small but significant and repeatable delay in flower initiation and flowering
time, and a lower flower/leaf relativity value (not significant) in aero2 plants (Table 1). There was also no evidence that the
photoperiod response was reduced in aero2
plants (data not shown).
The difference between the effects of the aero1
and aero2 mutations is interesting.
The AERO1 gene seems to be part of a
basic timing or clock mechanism where mutation leads to a general acceleration
of a diverse range of developmental processes including flowering (12). In
contrast, the effects of the aero2
mutation are more limited and do not extend to acceleration of flowering, a key
developmental process. This may mean that AERO2
acts downstream of AERO1 and with a
role in the timing of a more limited range of developmental events.
Acknowledgments:
We thank the Australian Research Council for financial support, Dr Jim Weller
for providing seed of the mutant, Dr Rob Wiltshire for the digital images used
in Figs 2 and 3, and Ian Cummings and Tracey Jackson for technical assistance.
SAT was supported by an Australian Postgraduate Scholarship.
1. Arney, S.E. and
Mancinelli, P. 1966. New
Phytol. 65: 161-175.
2. Blixt, S. 1962.
Agri Hort. Genet. 20: 95-110.
3. Gorel, F.L.,
Berdnikov, V.A. and Kosterin, O.E. 2002.
Pisum Genetics 34: 1-2.
4. Lu, B., Villani, P.J.,
Watson, J.C., DeMason, D.A. and Cooke, T.J. 1996.
Intl. J. Plant Sci. 157: 659-673.
5. Marx, G.A.
1986. Pisum Newslett. 18:
42-44.
6. Murfet, I.C. 1982.
Crop Sci. 22: 923-926.
7. Murfet, I.C. 1985.
Ann. Bot. 55: 675-685.
8. Murfet, I.C.
1990. Pisum Newslett. 22:
32-37.
9. Prajapati, S. and Kumar,
S. 2001. J.
Biosci. 26: 607-625.
10.
Sidorova, K.K. and Uzhintseva, L.P. 1995.
Russ. J. Genet. 31: 1008-1009.
11.
Sifton, H.B. 1945.
Bot. Rev. 11: 108-143.
12.
Taylor
,
S.A.
and Murfet, I.C. 2003. Physiol.
Plant. 117: 100-107.
13.
Villani, P.J. and DeMason, D.A. 2000.
Ann. Bot. 85: 123-135.
14.
Young, J.P.W. 1983.
Ann. Bot. 52: 311-316.
 Following recognition of the interesting and very strong pleiotropic
phenotype of aero1 (10, 12), we made a
detailed phenotypic comparison between WT and aero2
segregates in the F2 of the first and second backcrosses between the
mutant and cv. Torsdag. Both backcrosses gave consistent results. All plants
were grown in the glasshouse at Hobart in individual 14-cm slimline pots under an 18-h photoperiod. Further details on
growing conditions are as described earlier (12).
Following recognition of the interesting and very strong pleiotropic
phenotype of aero1 (10, 12), we made a
detailed phenotypic comparison between WT and aero2
segregates in the F2 of the first and second backcrosses between the
mutant and cv. Torsdag. Both backcrosses gave consistent results. All plants
were grown in the glasshouse at Hobart in individual 14-cm slimline pots under an 18-h photoperiod. Further details on
growing conditions are as described earlier (12).



