|
Pisum Genetics |
Volume 23 |
1991 |
Research Reports |
pages 44-49 |
The response to light quality in a yellow-green Pisum mutant
|
Weller, J.L. and Reid, J.B. |
Department of Plant Science, University of Tasmania Hobart, Tasmania 7001, Australia |
Important evidence for the existence of functionally differentiated pools of phytochrome has come from the study of mutants deficient in phytochrome or showing altered responses to phytochrome. Mutants au in tomato (11) and hy-1 and hy-6 in Arabidopsis (6, 12) are deficient in "etiolated-tissue" or "light-labile" phytochrome. These mutants lack some phytochrome responses (e.g. germination, photocontrol of stem elongation and cab expression in de-etiolating seedlings) but retain the phytochrome-mediated end-of-day far-red (EOD-FR) response. However, mutants lh in cucumber (2, 3) and Iv in pea (15) both lack the EOD-FR response, but retain phytochrome control of stem elongation in de-etiolating plants which the au, hy-1 and hy-6 mutants lack. Thus mutants lh and lv appear to have normal responses to light-labile phytochrome but seem to be deficient in response to a "light-stable" or "green tissue" phytochrome. The current hypothesis (see 8, 20) tentatively identifies labile phytochrome with the molecular species of phytochrome designated type I (PI) and stable phytochrome with type II phytochrome (PII). It is therefore considered that au, hy-1 and hy-6 are deficient in PI, while lh and lv are transduction chain mutants for PII. According to this theory, labile phytochrome (=PI?) controls the transition from dark to light, including seed germination and development of the photosynthetic apparatus (4, 14) and stable phytochrome (=PII?) controls shade avoidance-associated responses in the green plant such as stem elongation, apical dominance, and leaf morphology (13).
Conclusive identification of the two known physiological pools of phytochrome with specific molecular forms of the photoreceptor will require both appropriate phytochrome-deficient mutants, and the ability to distinguish between PI and PII. With the development of antibodies specific for pea PI and PII (1) and the recent sequencing of dicot phyB genes (7, 17) it is now possible to distinguish between the two different molecular forms of phytochrome in pea at both the protein and nucleic acid levels. Pea phytochrome mutants are therefore potentially of great value in the investigation of the developmental roles of the different forms of phytochrome. Recently, a yellow-green pea mutant, E158, was identified which also showed an apparent increase in elongation over the early internodes, and therefore appeared superficially similar to the au mutant in tomato. It was therefore of interest to examine E158 for evidence of altered phytochrome responses. To this end, we have characterized chlorophyll levels and the elongation response in E158 plants under several different light qualities.
Materials and Methods
The mutant E158 was derived by EMS mutagenesis from cv. Sparkle by Dr. T.A. LaRue of the Boyce Thompson Institute, Ithaca, N.Y., U.S.A. Both lines have internode length genes le, Lm, Ls, La, cryc, Na, Lh, Lv, Lw, Lk, Lka, Lkb and Lkc. Plants were grown as indicated in Reid and Ross (16). To examine the genetic control of the yellow-green phenotype in E158 the F2 of cross Sparkle x E158 was grown under an 18 h photoperiod in a heated glasshouse. Internode lengths of all plants were recorded at maturity, with internodes counted from the cotyledons as node 0.
The elongation response of E158 to light quality was examined by growing plants under continuous white, blue, red and far-red light, and in the dark. Growing conditions and light sources used were identical with those used by Nagatani et al. (15) with the exception of the white light intensity which was 6 mmol m-2 s-1. The total height of each plant (from soil surface to apical meristem) was measured at 8, 11, 14, and 18 days after sowing, with the exception of plants under far-red light and in the dark, which were not measured on days 8 and 11 to avoid exposure to light. These plants were measured under a green safety light on day 14. Plants were harvested 18 days after sowing, at which stage most plants had 3-4 internodes fully expanded. The total length (from cotyledonary node to the apical meristem) and the length of each fully expanded internode were recorded for each plant. Four plants of each line from each of the white, blue and red light treatment groups were frozen in liquid nitrogen and retained for chlorophyll analysis. Plant material for extraction consisted of that portion of the plant above node 2, amounting to approximately 0.8-1.0g of fresh tissue per plant. Chlorophyll was extracted and quantified following the method of Koornneef et al. (11).
Results
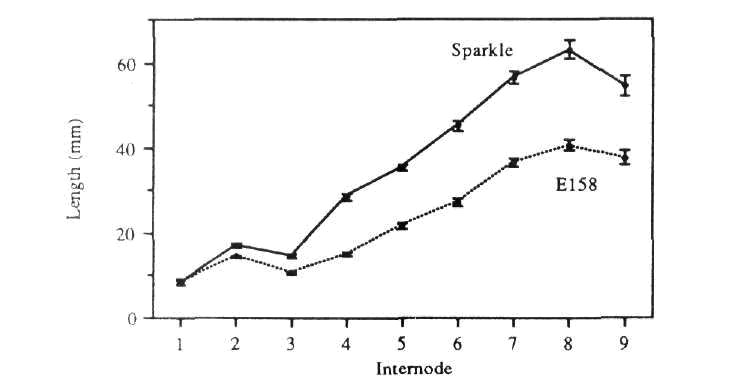
Glasshouse-grown E158 plants were significantly shorter than Sparkle plants over all internodes except internode 0-1 (epicotyl) which was the same length in both lines (Fig. 1).

Fig. 1. Comparison of internode lengths in Sparkle and E158 plants
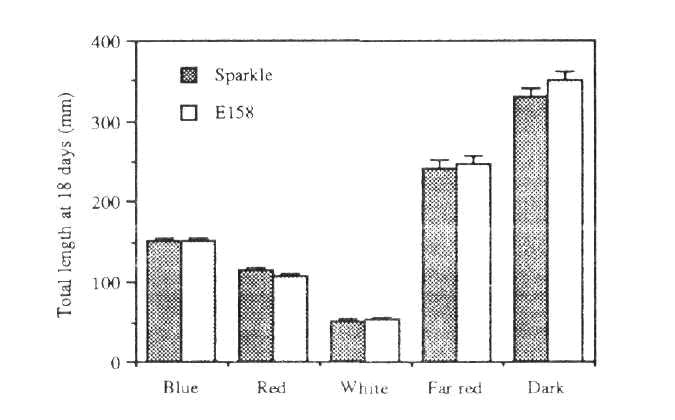
Fig. 2. Segregation for internode length in the F, of cross Sparkle x E158
Segregation in the F2 of the Sparkle x E158 cross was easily determined on the basis of foliage colour 1-2 days after emergence. In the F2 of 90 plants, 69 had normal foliage and the rest showed the yellow-green trait, in close accord with a 3:1 ratio (c2 = 0.13, P>0.5). Segregation was also observed in internode length (Fig. 2), with all yellow-green segregates also having shorter internodes between nodes 6 and 9 than segregates with normal foliage, thus suggesting that both traits result from a single recessive mutation.
At 18 days after sowing there was no significant difference in total length between E158 and Sparkle plants under any of the different light qualities (Fig. 3). Under all light qualities, at least some internodes were slightly longer in E158 than in Sparkle (Table 1). This difference was seen in internodes 1 and 2 under red and blue light, and in internodes 2 and 3 under white and far-red light, and in the dark. In addition, temporal data (not presented) showed that under white and blue light, E158 was slightly longer than Sparkle at 8, 11 and 14 days following emergence. Under red light there was no significant difference at any of the times measured.
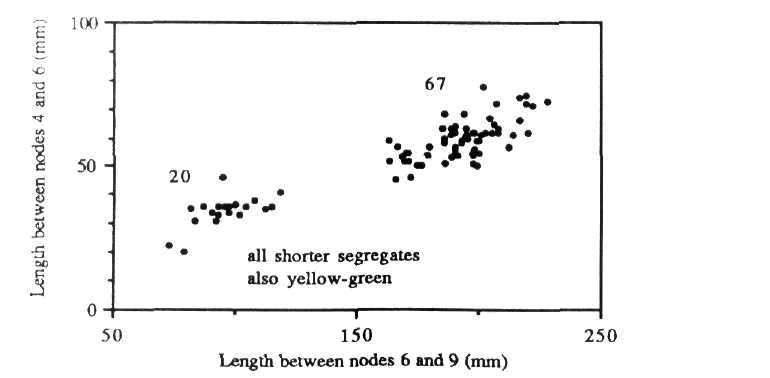
Fig. 3. Total lengths (from soil surface to apical meristem) of 18-day-old Sparkle and E158 seedlings grown under white, red, blue or far-red light or in darkness.
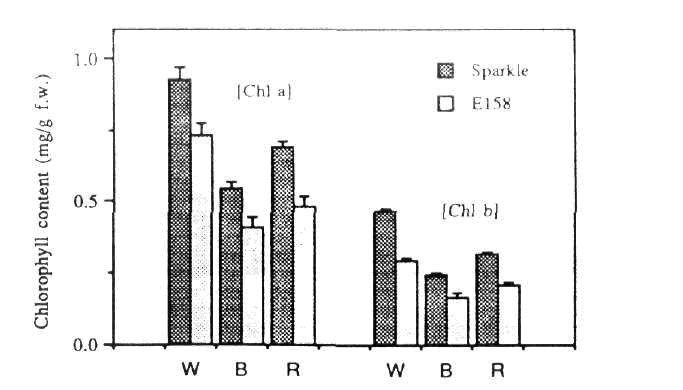
Fig. 4. Chlorophyll a and b levels in 18-day-old Sparkle and E158 seedlings grown under white, blue or red light.
Under all light treatments tested (i.e. blue, red and white) chlorophyll a and b levels (and thus total chlorophyll levels) in E158 were reduced by approximately 30% relative to levels in Sparkle (Fig. 4). A small (<10%) but significant increase in the chlorophyll a:b ratio was detected in E158 plants grown under blue and white light (Fig. 5).
Discussion
Photomorphogenic mutants au in tomato and hy-1 in Arabidopsis, which have both been suggested to involve phytochrome I, both show a substantial reduction in red light inhibition of stem elongation. This reduction in inhibition has been quantified in different ways for the two mutants, but in each instance the increase in stem length is at least two times greater than in the corresponding wild type (2,12). After 18 days under red light, E158 did not differ significantly in total length from the isogenic wild type Sparkle, and examination of the elongation response of the mutant under other wavelengths and in the dark provided no substantial evidence to suggest that E158 lacked phytochrome control of stem elongation. In order to address the question of whether E158 seedlings might only show reduced photoinhibition of elongation at a very early stage (i.e. prior to 18 days after emergence), the ontogenetic (Table 1) and temporal expression of the E158 / Sparkle difference was examined. Results showed that E158 was slightly longer than Sparkle in some internodes and in total length over the first few days following emergence. The reason for these differences is not clear. However, differences were seen under all light conditions, and thus did not seem to be directly dependent on light quality.
Table 1. Lengths (in mm) of intemodes 0-1, 1-2 and 2-3 in 18-day-old E158 and Sparkle plants grown under white, red, blue and far-red light and in darkness. The significance levels of differences between the two lines are indicated (**P<0.01, *P<0.05, NS P>0.05)
|
|
|
Length 0-1 |
Length 1-2 |
Length 2-3 |
||||||
|
|
|
E158 |
|
Sparkle |
E158 |
|
Sparkle |
E158 |
|
Sparkle |
|
|
mean |
9.44 |
|
9.22 |
7.56 |
|
5.00 |
16.78 |
|
13.89 |
|
White |
S.E. |
0.73 |
|
0.28 |
0.90 |
|
0.47 |
0.98 |
|
0.42 |
|
|
|
|
NS |
|
|
* |
|
|
* |
|
|
|
mean |
23.80 |
|
19.40 |
21.90 |
|
17.20 |
32.40 |
|
33.90 |
|
Red |
S.E. |
1.52 |
|
0.86 |
1.79 |
|
1.07 |
1.35 |
|
1.68 |
|
|
|
|
* |
|
|
* |
|
|
NS |
|
|
|
mean |
30.56 |
|
24.78 |
31.00 |
|
24.44 |
47.44 |
|
48.67 |
|
Blue |
S.E. |
2.77 |
|
1.34 |
2.15 |
|
1.79 |
1.52 |
|
1.04 |
|
|
|
|
NS |
|
|
* |
|
|
NS |
|
|
|
mean |
35.40 |
|
33.40 |
49.40 |
|
36.30 |
83.00 |
|
68.40 |
|
Far red |
S.E. |
2.74 |
|
1.59 |
3.19 |
|
1.69 |
3.89 |
|
2.90 |
|
|
|
|
NS |
|
|
** |
|
|
** |
|
|
|
mean |
65.11 |
|
61.90 |
70.78 |
|
54.00 |
148.00 |
|
109.80 |
|
Dark |
S.E. |
3.37 |
|
2.39 |
3.31 |
|
1.86 |
7.41 |
|
2.75 |
|
|
|
|
NS |
|
|
** |
|
|
** |
|
Although statistically significant, these differences are not of the order of elongation differences seen in established phytochrome mutants. Thus it appears that that the mutation in E158 has no substantial effect on red light inhibition of stem elongation. In this sense E158 differs from the apparently PI deficient au mutant. For this reason, it seems probable that the E158 mutation does not directly involve phytochrome, or phytochrome signal transduction. The reduction in internode length over later internodes seen in E158 plants grown to maturity under an 18 h photoperiod (Fig. 1) is most likely attributable to a decreased photosynthetic capacity resulting from the low chlorophyll content.
Further evidence that phytochrome is not directly involved in E158 was provided by examination of chlorophyll levels in the mutant. Expression of the gene encoding the chlorophyll a/b binding (cab) protein in pea is known to be under very sensitive phytochrome control (10), and accumulation of the cab protein in response to red light is an integral part of the de-etiolation process. As chlorophyll b is bound exclusively to the cab protein (9), an increased chlorophyll a:b ratio is therefore characteristic of plants which for some reason lack this protein. The phytochrome deficient mutant au is almost completely lacking in red light induction of cab gene expression (18) and cab protein accumulation, and consequently shows a substantial increase in the chlorophyll a:b ratio (11). In E158, chlorophyll levels were reduced by approximately 30% compared to Sparkle, but this represented an almost equivalent reduction in both chlorophyll a and chlorophyll b levels. The chlorophyll a:b ratio in E158 grown under white light was slightly increased (by about 10%) relative to Sparkle, but this increase was much less than increases seen in the other putative PI mutants au , hy-1 and hy-6, of 74%, 75% and 220% respectively. Mutant hy-2, which contains about 30% of the normal level of phytochrome, has a similar phenotype to E158 with respect to chlorophyll accumulation (70% of wild type chlorophyll content, chlorophyll a:b ratio of 1.18 x wild type) but shows markedly reduced inhibition of elongation under red light.
When considered together, the elongation and chlorophyll results strongly suggest that E158 is not a phytochrome I mutant. It is also unlikely that E158 is either a chlorophyll b mutant or a cab mutant as in both cases a substantial increase in the chlorophyll a:b ratio would be expected. Chlorophyll accumulation is a complex process and includes synthesis of chlorophyll, light-harvesting complex (LHC) assembly (which requires the synthesis of several proteins), and incorporation of chlorophyll into the LHC.
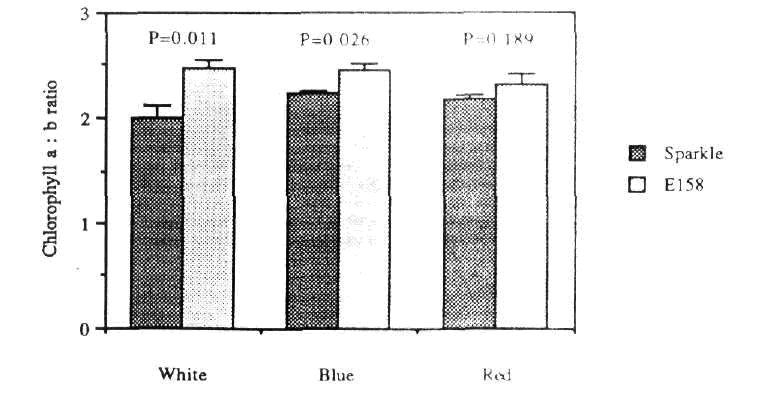
Fig. 5. Chlorophyll a:b ratio in Sparkle and E158 plants grown under white, blue or red light.
Thus, the reduction in chlorophyll levels seen in a mutant such as E158 may result from disruption of any of these processes.
White light grown E158 plants are characterized by reduced chlorophyll a and b levels, reduced plant height and reduced seed yield. On the basis of these characteristics E158 is best described as a mutant of the chlorotica type (see 5). This phenotype is known to result from mutation at any one of more than 20 loci. Severity of chlorosis varies widely among chlorotica mutants, with chlorophyll levels reported to range from 14% to 100% of wild type levels. Wide variation in chlorophyll a:b ratio is also reported, from 90% to 270% of the wild type ratio (see 5, 19).
Acknowledgements. We thank Dr T.A. LaRue for providing seed of E158 and the Australian Research Council for financial support.
Abe,
H., Yamamoto, K.T., Nagatani, A. and Furuya, M. 1985. Plant Cell
Physiol. 26(7): 1387-1399.
Adamse,
P., Jaspers, P.A.P.M., Kendrick, R.E. and Koornneef, M. 1987. J.
Plant Physiol. 127: 481-491.
Adamse,
P., Jaspers, P.A.P.M., Bakker, J.A., Kendrick, R.E. and Koornneef,
M. 1988. Plant Physiol. 87: 264-268.
Adamse,
P., Jaspers, P.A.P.M., Bakker, J.A., Wesselius, J.C, Heeringa,
G.H., Kendrick, R.E. and Koornneef, M. 1988. J. Plant Physiol. 133: 436-
440.
Blixt, S. 1972. Agri Hort. Genet. 30: 1-293.
Chory,
J., Peto, C.A., Ashbaugh, M., Saganich, R., Pratt, L.H., and Ausubel,
F. 1989. Plant Cell 1: 867-880.
Dehesh,
K., Tepperman, J., Christensen, A.H. and Quail, P.H. 1991. Mol.
Gen. Genet. 225: 305-313.
Furuya,
M. 1989. Molecular properties and biogenesis of phytochrome I and
II. Adv. Biophys. 25: 133-167.
Kaplan,
S. and Arntzen, C.J. 1982. In Photosynthesis: Energy Conversion
by Plants and Bacteria, Academic Press, New York, pp 65-140.
Kaufman,
L.S., Thompson, W.F., and Briggs, W.R. 1984. Science 226:
1447-1449.
Koornneef,
M., Cone, J.W., Dekens, R.G., O'Herne-Roberts, E.G., Spruit,
C.J.P. and Kendrick, R.E. 1985. J. Plant Physiol. 120: 153-165.
Koornneef,
M, Rolff, E., and Spruit, C.J.P. 1980. Z. Pflanzenphysiol. 100:
147-160.
Lopez-Juez,
E., Buurmeijer, W.F., Heeringa, G.H., Kendrick, R.E. and
Wesselius, J.C. 1990. Photochem. Photobiol. 52: 143-149.
Lopez-Juez,
E., Nagatani, A., Buurmeijer, W.F., Peters, J.L., Furuya, M.,
Kendrick, R.E. and Wesselius, J.C. 1990. J. Photochem. Photobiol., B:
Biology 4: 391-405.
Nagatani,
A., Reid, J.B., Ross, J.J., Dunnewijk, A. and Furuya, M. 1990. J.
Plant Physiol. 135: 667-674.
Reid, J.B. and Ross, J.J. 1988. Physiol. Plant. 72: 595-604.
Sharock, R.A. and Quail, P.H. 1989. Genes Dev. 3: 1745-1757.
Sharrock,
R.A., Parks, B.M., Koornneef, M. and Quail, P.H. 1988.
Molecular analysis of the phytochrome deficiency in an aurea mutant of
tomato.
Mol Gen. Genet. 213: 9-14.
Stummann, B.M. and Henningsen, K.W. 1980. Hereditas 93: 261-275.
Tomizawa,
K.-i., Nagatani, A. and Furuya, M. 1990. Photochem. Photobiol.
52: 265-275.