|
PNL |
Volume 22 |
1990 |
RESEARCH REPORTS |
pages 41-42 |
EVIDENCE OF MATERNAL TRANSMISSION OP PLASTID DNA IN THE GARDEN PEA
|
Polans, N.O,
J.L. Corriveau
and A.W. Coleman
|
Department of Biological Sciences, Northern Illinois University, DeKalb, IL 60115, USA Department of Agronomy, University of Maryland College Park, MD 20742, USA Division of Biology and Medicine, Brown University Providence, RI 02912, USA |
Plastids are inherited maternally in most angiosperm species, although an increasing number of exceptions to this rule are being discovered (8-10). The mode of plastid inheritance in pea remains unresolved owing to the contradictory findings of both genetical and ultrastructural studies. DeHaan (5) originally reported that a chlorophyll deficiency was inherited maternally in Pisum sativum L. His subsequent report, however, specified the biparental inheritance of the same variegation in a small number of crosses (6). We responded to these observations by emphasizing the need to determine whether the chlorophyll deficiency trait was a nuclear-controlled plastid deficiency or a plastid DNA mutation (4). We also suggested that DeHaan may have worked initially with line(s) following the maternal mode of plastid transmission and only later with line(s) capable of occasional biparentalism. Evidence of variability for mode of plastid DNA inheritance has been reported for Pelargonium (11), Oenothera (2), and Petunia (3).
In ultrastructural studies of pea microgametogenesis, Biddle (1) reported the absence of plastids in the generative cells of the pollen grains she examined, thereby suggesting the implausability of paternal plastid transmission. Hause (7), however, reported the presence of a small number of proplastids in the generative cells of pollen grains from cv Fruhe Harzerin, reintroducing the possibility of paternal plastid transmission. The discrepancy between these studies might be explained, once again, by variability among pea lines for mode of plastid inheritance.
In order to determine whether there might be variability among pea lines for paternal plastid transmission, we examined 13 lines cytologically using the DNA-fluorochrome DAPI to quantify levels of DAPI-stained DNA nucleoids in the generative cells of mature pollen (4). We detected plastid DNA in all the lines examined, although there was variability among lines with regard to both the percentage of pollen grains containing plastid nucleoids (3% to 65%) and the number of nucleoids per generative cell (none to 18). These results suggested genetic variability for this characteristic in pea, as well as the possibility that P. sativum might sometimes exhibit biparental inheritance of plastids, particularly in those paternal lines displaying high levels of pollen plastid DNA. At issue was whether paternal plastid transmission was correlated with the availability of plastids in the pollen.
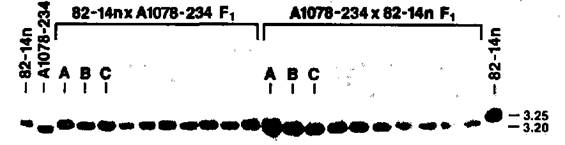
We have addressed this question by using (chloroplast) RFLP markers to follow the transmission of plastid DNA from parents to offspring. Crosses were designed so that maternal and paternal plastids could be differentiated unambiguously on the basis of length variation in a single plastid DNA fragment. Each parent produced either a 3.20 kb, 3.25 kb or 3.35 kb XhoI band corresponding to the "hypervariable" region in the pea plastid genome. The data from a series of crosses clearly indicated only the presence of the maternal band in the F1 progeny of each cross examined, irrespective of the amount of plastid DNA detected cytologically in the pollen of the paternal parent. When reciprocal crosses were specifically constructed to maximize the differences in plastid DNA content between parents possessing different RFLP markers, the same results were obtained (see, e.g., Fig. 1), even when F1 progeny were sampled at early developmental stages, putatively prior to any "sorting-out" of plastid types. Thus, P. sativum appears to follow a uniparental-maternal mode of plastid inheritance; although, the number of F1 progeny examined in each cross was too small to eliminate the possibility of trace biparentalism in pea. Furthermore, there does not appear to be an obvious correspondence between the presence of plastid DNA in the generative cells of germinated pea pollen and its subsequent transmission to F1 offspring.
Biddle, J.A. 1979. Can. J. Bot. 57:1883-1900.
Chiu, W.-L., W. stubbe and B.B. Sears. 1988. Curr. Genet. 13:181-189.
Cornu, A. and H. Dulieu. 1988. J. Hered. 79:40-44.
Corriveau, J.L., N.O. Polans and A.W. Coleman. 1989. Curr. Genet. 16:47-51.
DeHaan, H. 1930. Genetica 12:321-440.
DeHaan, H. 1933. Bibliogr. Genet. 10:357-416.
Hause, G. 1986. Biol. Zentralbl. 105:283-288.
Sears, B.B. 1980. Plasmid 4:233-255.
Smith, S.E. 1988. Plant Breed. Rev. 6:361-393.
Tilney-Bassett, R.A.E. 1978. In The Plastids, Eds J.T.O. Kirk and R.A.E. Tilney-Bassett, Elsevier/North-Holland, Amsterdam, pp. 251-524.
Tilney-Bassett, R.A.E. and O.A.L. Abdel-Wahab. 1979. In Maternal Effects in Development, Eds D.R. Newth and M. Balls, Cambridge Univ. Press, London, pp. 29-45.

Fig. 1. Auto radiograph of pea parental and F1 progeny DNA. XhoI band fragments approximately 3.20 and 3.25 kb in length are revealed in representative samples from the reciprocal cross between line 82-14n (65% of pollen generative cells contain plastid DNA, 3.25 kb XhoI marker band) and line A1078-234 (16%, 3.20 kb) after hybridization with a 2.3 kb mung bean probe (kindly provided by J.D. Palmer). Even when the film is overexposed, only the maternal band variant is observed in the F1 progeny. Maternal plants are listed first in each cross. The letters A-C represent leaf material collected from a mixture of young F1 plants at successive five-day intervals. Unmarked lanes represent individual mature plants. The relatively diffuse band in Lane A of the A1078-234 x 82-14n cross originally suggested the possible presence of the paternal band fragment. However, additional examination of this DNA confirmed the presence of only the maternal (3.20 kb) band fragment.