
|
PNL |
Volume 22 |
1990 |
RESEARCH REPORTS |
pages 32-37 |
WATER CONGESTION DAMAGE IN PEA: REDUCED SUSCEPTIBILITY IN THE SMALL STIPULE MUTANT, st
|
Murfet, Ian C.
|
Department of Plant Science, University of Tasmania Hobart, Tasmania 7001, Australia |
After extensive trials, Johnson (4) placed the garden pea among those species highly susceptible to water congestion. Hagedorn and Rand (1, 2) reported that water congestion is sometimes a troublesome malady in the processing pea crop and although considerable diversity in degree of susceptibility occurred among 133 processing pea cultivars examined, none was found wholly free from the malady. They described the condition as characterised by a dying back of the terminal and outer edges of the pea leaflets and stipules. In severely afflicted plants up to 75% of the foliage area at several nodes was destroyed. No pathogen was associated with this disease which appeared most commonly under conditions of high soil moisture, high relative humidity and warm temperatures. Water congestion also occurs in plants grown in our greenhouse and phytotron facility at Hobart (J.J. Ross unpub.). The problem is particularly troublesome in our flowering studies where the plants are sometimes held for several months in short day conditions. In these circumstances severely afflicted plants may lose over 90% of leaflet area at some nodes (Fig. 1) and in extreme cases the apex may be destroyed. In a recent flowering study involving a cross between Hobart lines 67 and 210 some very marked differences in the pattern of water congestion damage occurred among F2 segregates. The reduction in susceptibility of segregates homozygous for the recessive allele st conferring small stipules (6) was particularly striking (Fig. 2) but effects of daylength and the onset of fruiting were also clearly apparent. I have therefore presented here data which illustrate the effect of these three factors on the expression of water congestion in peas.
Materials and methods
The data in Figs 3-5 come from F2 and F3 plants of a cross between pure breeding Hobart pea lines 67 (normal stipules, genotype St/St) and 210 (small stipules, st/st). Plants of both generations were sown at the same time. Individual plants were grown in 14 cm slim line pots in a 1:1 mixture of vermiculite and dolerite chips (6 mm) topped with 4 cm of sterilized peat/sand potting mix. All plants received 8 h daylight in the glasshouse at about 20°C followed by 16 h in growth chambers at 16ºC, either in darkness (8 h photoperiod) or under lights for 6 h (14 h photoperiod) or 16 h (24 h photoperiod). Lighting in the growth chambers was provided by Thorn 40 W white fluorescent tubes (photon flux 50 mmol m-2 s-1 at pot top) and Mazda 40 W incandescent globes (5 mmol m-2 s-1). Nutrient in the form of Hoaglands solution was supplied once a week. Plants were watered to field capacity three times a week for the first four weeks and thereafter daily.
Leaf damage resulting from water congestion was rated visually on a seven point scale (see legend to Fig. 3). The most severe damage category, loss of the main-shoot apex, occurred only rarely and was always associated with massive damage to the leaflets. In such cases shoot growth continued through a lateral branch which was free of damage initially but displayed the same pattern of water congestion damage as the main shoot once it assumed the ascendency.

Fig. 1. A plant with normal stipules (St) showing symptoms of severe water congestion damage including almost complete loss of leaflet area in the case of the leaf top right. Lateral branches arising in the axils of these damaged leaves were initially healthy (see above) but subsequently developed severe damage in later formed leaves. This plant was grown in an 8 h photoperiod (see text).

Fig. 2. A plant with reduced stipules (st) grown in the same conditions as the St plant in Fig. 1 but free of water congestion damage.
The data in Figs 3-5 were obtained from 78-day-old plants from cross 67 x 210. In Fig. 3, the 24 h data were obtained from all F2 plants in this photoperiod and these plants were all flowering, while the 14 h data were obtained from Hr/- F2 segregates only and these plants were all wholly vegetative. [The flowering gene Hr confers a high response to photoperiod (5)]. In Fig. 4, the data are for F3 plants of genotype st/st Hr/- from 4 progenies each contributing about the same number of plants in each photoperiod; these F3 plants were all flowering in the 24 h photoperiod but wholly vegetative in the 14 h and 8 h photoperiods. The data in Fig. 5 were obtained on day 78 when the plants had some 31-35 expanded leaves. The data for the upper leaves were obtained from the top five mature leaves and the data for the lower leaves from all leaves below the top five. The upper five leaves all subtended flowers or developing fruits in the hr/hr segregates which commenced flowering 8-12 days prior to day 78. The Hr/- segregates were wholly vegetative on day 78.
Results
The data in Figs 3-5 show that stipule type, daylength and flower bud development all exerted a marked effect on expression of water congestion. Thus to examine the effect of one factor the other two must be held constant or at least taken into account.
The reduction in susceptibility to water congestion conferred by the presence of small stipules is clearly evident from a comparison of maximum leaf damage in st/st and St/- segregates of the 67 x 210 F2 (Fig. 3). No segregate with small stipules (st/st) exceeded a damage rating of 1 in either the 24 h or 14 h photoperiod. In contrast, the majority of plants with normal stipules (St/-) had a damage rating of 2 to 4. All F2 plants from the 24 h photoperiod were included in the comparison since all were flowering at the time of the survey. In the 14 h photoperiod the comparison was confined to segregates with flowering genotype Hr/- and these plants were all wholly vegetative at the time of scoring.
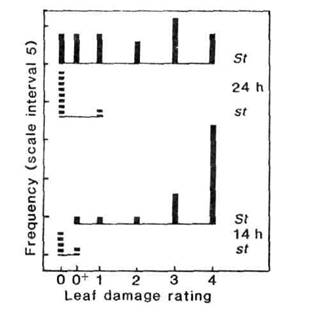
Fig. 3. Effect of stipule type on water congestion damage.
Maximum leaf damage rating for segregates with normal stipules ( ![]() ;
genotype St/-) or small stipules (
;
genotype St/-) or small stipules ( ![]() ;
st/st) in the F2 of cross 67 x 210 grown in a photoperiod of 24 h
(all plants flowering) or 14 h (Hr/-segregates only, all plants
vegetative). Damage rating scale: no visible damage (0); trace of damage (0+);
loss of leaflet area at the most severely afflicted leaf estimated as 1-25% (1),
26-50% (2), 51-75% (3), 76-100% (4); shoot apex destroyed (5).
;
st/st) in the F2 of cross 67 x 210 grown in a photoperiod of 24 h
(all plants flowering) or 14 h (Hr/-segregates only, all plants
vegetative). Damage rating scale: no visible damage (0); trace of damage (0+);
loss of leaflet area at the most severely afflicted leaf estimated as 1-25% (1),
26-50% (2), 51-75% (3), 76-100% (4); shoot apex destroyed (5).
The increasing severity of water congestion with decreasing photoperiod is illustrated in Fig. 4. Line 67 (normal stipules, genotype St/St) is one of the most susceptible lines in the Hobart collection. The leaf damage rating was 3 to 4 even in the 24 h photoperiod but in the 8 h photoperiod damage was so severe that the apex was destroyed in two of the four plants. The results for F3 plants of genotype st/st Hr/- show that while small stipules markedly reduced susceptibility to water congestion, quite significant leaf damage still occurred in conditions strongly conducive to expression of the malady and approximately one fifth of the st/st plants reached a maximum damage rating of 2 in the 8 h photoperiod. In contrast, the st/st plants showed no damage in the 24 h photoperiod.
The data in Fig. 5 suggest that susceptibility to water congestion is diminished by the onset of flowering and fruiting. The results come from F2 plants in 14 h conditions and involve a comparison of segregates which had normal stipules (St/-) but which differed in their flowering genotype at the Hr locus. The hr/hr segregates produced open flowers 8-12 days prior to collection of the water congestion data while the Hr/- segregates were all wholly vegetative at the time of scoring. The two flowering genotypes (corresponding to the flowering phenotypes L and LHR, respectively; see 5) both showed much the same leaf damage rating for lower leaves which expanded prior to the appearance of flowers in the hr/hr segregates. However, when the upper leaves were compared it was clear that leaves in hr/hr plants which expanded after the onset of flowering showed markedly less damage than equivalent leaves in the still vegetative Hr/- plants which expanded during the same time period.
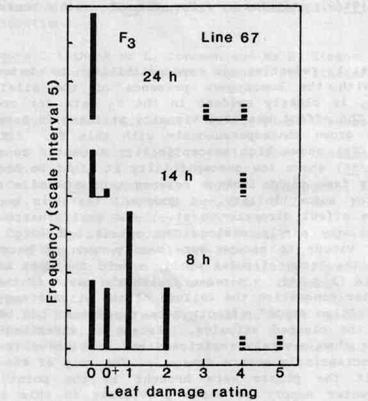
Fig. 4. Effect of photoperiod on water congestion damage.
Maximum leaf damage rating for F3 ( ![]() )
plants of genotype st/st Hr/- and line 67 plants (
)
plants of genotype st/st Hr/- and line 67 plants ( ![]() ;
St/St Hr/Hr) in photoperiods of 24 h (all plants
flowering), 14 h (all plants vegetative) and 8 h (all plants vegetative). For
damage rating scale see Fig. 3 caption.
;
St/St Hr/Hr) in photoperiods of 24 h (all plants
flowering), 14 h (all plants vegetative) and 8 h (all plants vegetative). For
damage rating scale see Fig. 3 caption.
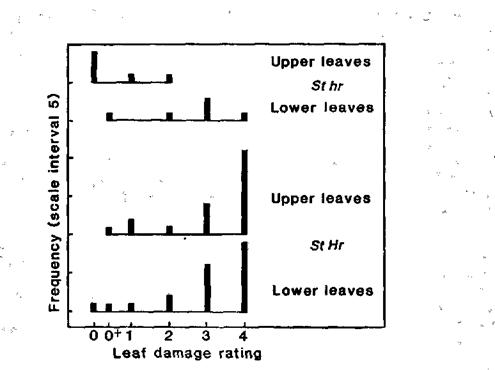
Fig. 5. Effect of flowering on water congestion damage. Maximum leaf damage rating for the upper (top 5) and lower leaves on 78-day-old F2 plants of cross 67 x 210 which had either commenced flowering 8-12 days prior to day 78 (genotype hr/hr) or which were still wholly vegetative on day 78 (genotype Hr/-). Photoperiod 14 h; all plants had normal (St/-) stipules. For damage rating scale see Fig. 3 caption.
Discussion
The substantial reduction in susceptibility to water congestion associated with the homozygous presence of the allele for small stipules, st, is clearly evident in the F2 data for cross 67 x 210 (Fig. 3). The effect was also visually striking in several 67 x 210 F3 progenies grown contemporaneously with this F2. Since line 67 (genotype St/St) shows high susceptibility to water congestion while line 210 (st/st) shows low susceptibility it might be argued that the effect arises from tight linkage between gene st and a separate gene conferring low susceptibility. However, there is good reason to attribute the effect directly to st. The small, narrow stipules in st/st plants only partly enclose the apical bud (Fig. 2) and this allows water vapour to escape more easily than is possible in St/-plants where the large stipules wholly enfold the apex and developing leaf primordia (Fig. 1). Moreover, with the onset of the more severe stages of water congestion the failure of the stipule margin to expand results in a "clam shell" effect where the apical bud becomes wholly enclosed by the clamped stipules. Escape of water vapour from the apical bud is then severely restricted and a positive feedback effect results in increasingly severe damage. The grip of the malady could be broken if the plants were brought to the point of wilt by withholding water supply as shown previously in this laboratory by J.J. Ross (unpub.).
The reduction in water congestion following the onset of flowering (Fig. 5) may likewise occur because the swelling flower buds hold open the array of developing stipules so allowing better ventilation of the apical bud. However, I have noticed that plants with developing fruits appear to demand more water than similar sized vegetative plants and it is possible that the hr/hr (flowering) segregates in Fig. 5 were, on average, closer to wilt-point than the Hr/- (vegetative) segregates. Both types were watered to field capacity each morning. The physiological basis for the increased expression of water congestion in short day conditions (Fig. 4) is not clear but the effect was also observed in a more detailed study in this laboratory by D.J. Bolton (unpub.).
Allele st offers scope for reducing susceptibility to water congestion. However, homozygosity for st itself results in a considerable reduction in photosynthetic area since stipule area is reduced to 8-14% of normal and total leaf area to around 68% of normal (10). Some reduction in dry seed yield in st/st compared with normal types was observed in field tests (9,10). Nevertheless, shelled pea yield corrected to 100 tenderometer was fairly similar in st/st and normal plants (10). Moreover, planting density is important in any evaluation of genotypes with reduced light interception (3) and some loss of photosynthetic area may be tolerated if it is offset by other benefits, e.g. the afila (af) mutant replaces all leaflet sites with branched tendrils but confers advantages such as improved standing ability (3,7-10). The effects of water congestion extend beyond foliar damage since it may cause the anthers to shrivel without dehiscing and hence lead to failure of seed set. Likewise, st also influences several other important traits such as vine length, tillering and blonding of seed (9,10) and it may also confer increased susceptibility to downy mildew (H. Brotherton, T.L. Lund and F.J. Muehlbauer pers. com.). The net effect of st can only be evaluated by field trials in appropriate locations.
Acknowledgements. I thank Mr L. Johnson and Ms D. Steane for technical assistance and the Australian Research Council for financial support.
Hagedorn, D.J., and R.E. Rand. 1971. Plant Disease Reporter 55:249-253.
Hagedorn, D.J., and R.E. Rand. 1971. Plant Disease Reporter 55:533-535.
Hedley, C.L. and M.J. Ambrose. 1981. Adv. Agron. 34:225-277.
Johnson, J. 1947. Wisconsin Agr. Exp. Sta. Res. Bull. 160:1-34.
Murfet, I.C. 1973. Heredity 31:157-164.
Pellew, C. and A. Sverdrup. 1923. J. Genet. 13:125-131.
Snoad, B. 1974. Euphytica 23:257-265.
Snoad, B. 1985. In The pea crop. A basis for improvement. Eds P.D. Hebblethwaite, M.C. Heath and T.C.K. Dawkins. Butterworths, London, pp 31-41.
Snoad, B., L. Frusciante and L.M. Monti. 1985. Theor. Appl. Genet. 70:322-329.
Wehner, T.C. and E.T. Gritton. 1981. J. Amer. Soc. Hort. Sci. 106:272-278.