PNL Volume 19 1987 RESEARCH
REPORTS
91
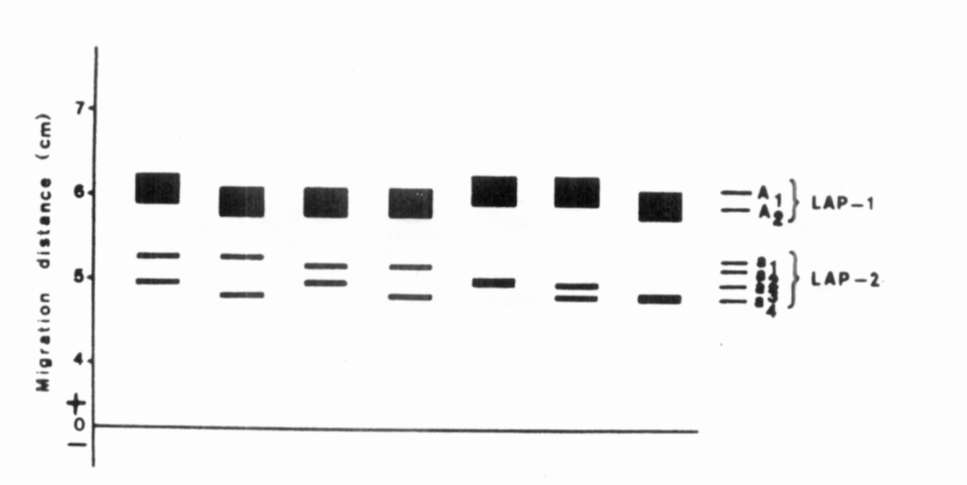
Fig. 1. The range of enzymatic variation of
LAP.
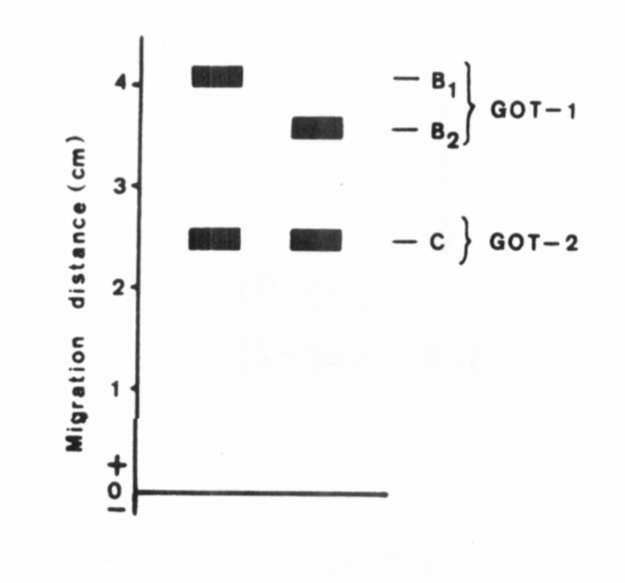
Fig. 2.
The range of enzymatic
variation of GOT.
variation of GOT.
|
|
||
|
PNL Volume 19 1987 RESEARCH
REPORTS 89 |
||
|
|
||
|
APPLICATION OF ELECTROPHORETIC
METHODS OF ISOZYME SEPARATION
TO GENETICAL CHARACTERIZATION OF PEA CULTIVARS Wolko, B. and W. K. Swiecicki
Plant Breeding Station, Wiatrowo, Poland |
||
|
|
||
|
Determination of varietal
distinctness, homogeneity, and sta-
bility (DHS) often proves difficult for individuals and agencies responsible for providing variety descriptions. The descriptions of varieties
include many imprecise, poly-
genic features, strongly influenced by the environment (e.g. the hue of green of field-grown plants or classes of stem length [e.g. tall, medium, short]). Suitable markers for distinguishing doubt- ful cases are a decided advantage. The most valuable are mono- genic features with stable expression and full penetrance, so- called variety markers. Electrophoretic methods of
isozyme separation are highly
suitable for this purpose. In Pisum the studies of Przybylska et al. on isozymes of different taxa were important (2). Significant also were studies of Weeden (3,4,5). The main aim of our work was to study the range of enzyme variation of pea cultivars in three enzyme systems controlled by at least six loci and to demonstrate the usefulness of isozyme analyses for cultivar characterization. Fifty-six cultivars from the
Wiatrowo collection were inves-
tigated, including cultivars having different uses (vegetable, dry edible seed, fodder, and green forage) bred and cultivated in Po- land as well as other countries. The group of cultivars initially
was characterized according
to morphological features (1). In exceptional cases it was possi- ble to distinguish the variety from the others without any doubts (e.g. only cv. 'Novella' has genotype af, fa) . But some varieties could not be distinguished using the hitherto known methods of description and identification. LAP, GOT (=AAT), and 6PGD
were investigated using Leaf tissue.
Electrophoretic separation was performed in starch gels (Serva) using well-known procedures. Electrophoretic phenotypes found among the cultivars analyzed are presented in Figs. 1, 2, and 3. Isozyme genes were useful for characterizing and distinguishing varieties in the following cases: 1. Cultivar of different origin with identical
phenotype.
Morphological criteria alone could not show a difference between cultivars 'Neuga' (DDR) and 'Oraniencroon' (ZAF). But in t lie locus Lap-1 cv. Neuga has the allele A1 and cv. Oraniencroon has A2. Moreover, polymorphism was observed in cv. Neuga for 6Pgd-2. We have designated this locus 6pgd-c. There were in- dividuals with the allele E1 (10%) and with E2 (90%). Therefore, Neuga and Oraniencroon, in spite of the same morpholo- gical phenotype, have different genotypes. They differ with re- gard to two monogenic characters, and thereby fulfill the require- ment for distinctness of cultivars. 2. Electrophoretic separation of isozymes can be
used for the description of selected varieties. For example, the Wiatrowo gene bank contains two cultivars named 'Wonder of Amerika', each |
||
|
|
||
|
|
|||
|
90 |
PNL Volume 19 1987 RESEARCH REPORTS |
||
|
|
|||
|
from a different country. They have
dissimilar electrophoretic
phenotypes in the zone of enzyme activity of LAP-2 and a different allele of the 6Pgd-2. The fact that both varieties are monomorphic for opposed alleles suggests that they possibly were selected from another, third line which was polymorphic for the gene 6Pgd-2. 3. It
is interesting to compare cultivars of close origin,
especially when they were bred from the same parental forms. For example, 'Aster' and 'Opal', bred in different breeding stations (Lipie and Wiatrowo), both originated from reciprocal crosses of cv. 'Auralia' and 'Flavanda'. Analysis of enzyme variation of the parental varieties showed polymorphism in Auralia for Lap-1 (allele A1 in 63% of the individuals, A2 in 37%) and Lap-2 (a1a3 in 47%, and a2a3 in 53%). Flavanda had the phenotype a1a3 of Lap-2 and the allele A1 of locus Lap-1. Moreover, this variety was polymorphic lor the locus 6Pgd-2 (D1 5% and D2 95%). Cultivars bred from crossing Auralia with Flavanda possessed the allele A1 of Lap-1 and the allele D1 of 6Pgd-2. However, they differ in phenotypes of Lap-2 (Opal a2a3; Aster - a1a3). Thus, Aster and Opal differ from each other as well as from the
parental varieties by at least one
allele. A similar analysis can be conducted
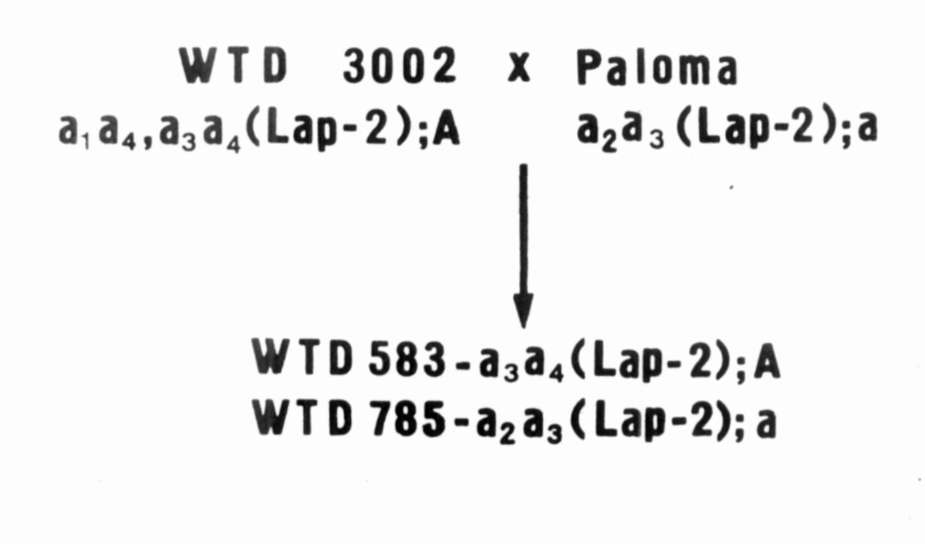
for strains WTD 583 and
WTD 785 originating from the cross combination WTD 3002 x 'Paloma'. Both parental forms as well as the breeding strains differ from each other by the electrophoretic pattern in the Lap-2 locus and have different alleles of gene A (anthocyanin synthesis) (Fig. 4). 4. Since cultivars are developed without regard to
enzyme
variation but, instead are selected for uniformity and for horti- cultural merit, it is of interest how much isozyme polymorphism developed varieties contain. For example, line Wt 4367 was poly- morphic for LAP-1, LAP-2, G0T-1, 6PGD-1 6PGD-2. In one population stable frequency of alleles of polymorphic genes was found in two consecutive years, 1984 and 1985. Polymorphism was also observed in 13 other cultivars. Thus, pea varieties, although pure lines with regard to a number of characters considered during the breed- ing process, can be polymorphic from the point of view of isozyme loci. The implications or importance of this is not known. Poly- morphism of enzyme variants is not an obstacle for variety identi- fication because a stable frequency of alleles of a given locus in a population characterizes a cultivar, too. The results presented above indicate
the possibilities of
employing electrophoresis for cultivar description. Together with a group of monogenic characters of stable expression, recognizable in seedlings and in seeds, electrophoresis should become a part of the routine tools used by institutions for registering varieties and testing seed, especially as a supplement when existing des- criptions used for establishing DHS are insufficient. 1.
Blixt, S. and W. K.
Swiecicki. 1980. Post. Nauk Rol. 6:31-38.
2. Przybylska, J., S. Blixt, H.
Parzysz, and Z. Zimniak-
Przybylska. 1982. Gen. Pol. 23:103-121. 3. Weeden, N. F. 1983. PNL 15:58-59.
4. Weeden, N. F. 1985. The Pea Crop. P. D.
Hebblethwaite,
M. C. Heath, and T. C. K.
Dawkins, Eds. Butterworths,London.
pp. 55-66. 5. N. F. Weeden and G. A. Marx.
1984. J. Hered. 75:365-370. |
|||
|
|
|||
|
|
||||
|
PNL Volume 19 1987 RESEARCH
REPORTS |
91 |
|||
|
|
||||
 |
||||
|
|
||||
|
Fig. 1. The range of enzymatic variation of
LAP. |
||||
|
|
||||
 |
Fig. 2.
The range of enzymatic variation of GOT. |
|||
|
|
||||
|
|
|||
|
92 |
PNL Volume 19
1987 RESEARCH
REPORTS |
||
|
|
|||
 |
|||
|
|
|||
|
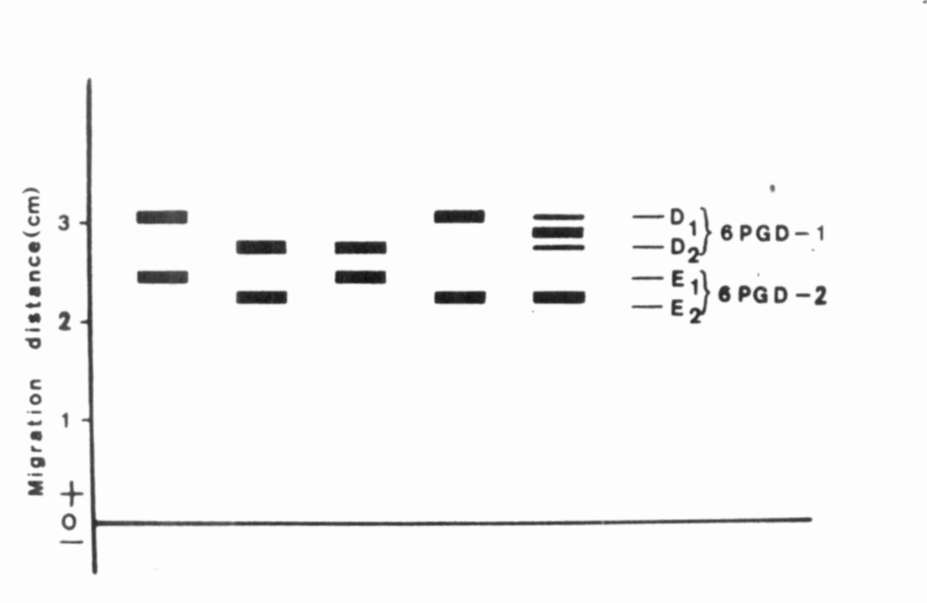
Fig. 3. The range of enzymatic variation of 6PGD. |
|||
|
|
|||
 |
|||
|
|
|||
|
Fig. 4. The origin and the genotype of the strains WTD 583 and WTD 785. |
|||
|
|
|||
|
***** |
|||
|
|
|||