14
PNL Volume 18 1986 RESEARCH REPORTS
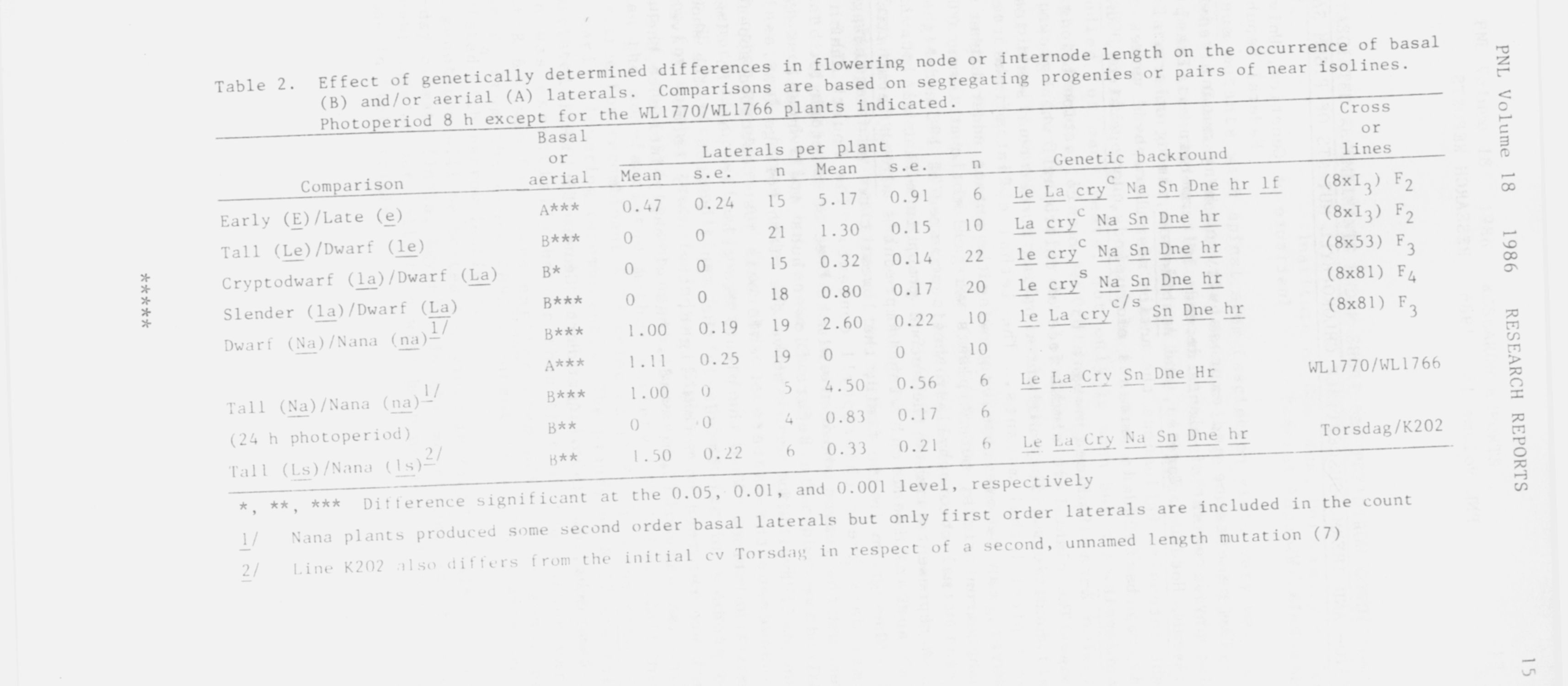
to poor growth since vigor in K202 was comparable with that of na nana
lines or to gene ls alone since a second length mutation is also present in
K202 (see 7). Moreover, the _ls type line, M26 (nana habit), shows profuse
basal branching (J. B. Reid, personal communication) in accord with the
pattern established for the na_ nana plants (Table 2).
No general trend was apparent for the effect of the length differences
on aerial laterals except for the paucity of such laterals in nana types
(Table 2).
We suggest that the length genes may influence branching in Pisum by
altering the level of available nutrients. One thing is certain, the
flowering and length genes have major effects on branching and the back-
ground for these genes and environmental factors such as photoperiod should
be taken into account in any study of branching per se although some genes
with specific effects on branching may be expressed regardless of these
factors.
1. Doroshenko, A. V. and V. [. Rasumov. 1929. Tr. Prikl. Bot. Oenet.
Sel. 22 :2 19-2 76.
2. King, W. M. and I. C. Murfet. 1985. Ann. Bot. 56:815-846.
3. Murfet, I. 1973. Heredity 31:157-164.
4. Murfet, I. 1985. Pp. 97-126 in Handbook of Flowering, Vol. IV. Ed.
A. H. Halevy, CRC Press.
5. Murfet, T. C. and J. B. Reid. 1974. Z. Pflanzenohysio1. 71:323-331.
6. Murfet, I. C. and J. B. Reid. 1985. pp. 67-80 in The Pea Crop: A
Basis for Improvement. Eds. P. D. Hebblethwaite, M. C. Heath, and T.
C. K. Dawki n s, Butter wo r t h s.
7. Reid, J. B. 1986. Ann. Bot. (in press).
8. Reid, J. B. and I. C. Murfet. 1984. Ann. Bot. 53:369-382.
9. Reid, J. B., 1. C. Murfet, and W. C. Potts. 1983. J. Exp. Bot.
34:349-364.
10. Ross, J. J. 1983. PhD Thesis, Univ. of Tasmania.
11. Ross, J. J, and 1. C. Murfet. 1985. Ann. Bot. 56:847-856.