RESEARCH REPORTS
PNL Volume 11
1979
TESTA ANTHOCYANINS IN PISUM
Crowden, R. K. University of Tasmania, Hobart, Australia
An earlier study of testa pigmentation in the Weibullsholm pea col-
lection (1) showed the occurrence of possibly nine anthocyanins. Although
only two pigments were fully identified at this time it was clear that the
b locus determined a qualitative difference in pigmentation: plants were
characterized by the presence of delphinidin derivatives and b plants by
the absence of delphinidin and the presence of cyanidin derivatives. The
results also indicated a quantitative relationship between certain genes
and anthocyanin production. No anthocyanins were detected in the absence
of gene A.
Six-of the nine pigments have now been identified. Pigments 3 and
7 [ numbered as in (1)] proved to be identical. The status of pigments 4
and 5 is unclear, but they are probably artifacts which arise sporadically
during the extraction process.
Testas of lines shown to be rich in specific anthocyanins or particular
mixtures of pigments were extracted in quantity, and the anthocyanins iso-
lated and purified using procedures described by Jarman and Crowden (2).
Structural determinations were then made on the rigorously purified pigments
using the standard procedures outlined by Harborne (3).
The identity of the pigments and their distribution in lines used for
this work are given in Table 1. Whereas various gene combinations involving
F, Fs, Ust, and U were seen to promote high anthocyanin production (1),
these" genes appear to bear no relationship to the distribution of pigments
among the different lines. Six distinct groupings emerge based on pigment
variation. Five groups (all B) contain delphinidin anthocyanins (supple-
mented by occasional trace amounts of cyanidin). Differentiation between
these groups is based on glycoside variation. The sixth group (b_) contains
cyanidin and pelargonidin anthocyanins, and relates directly to group 5
in terms of glycosidation pattern.
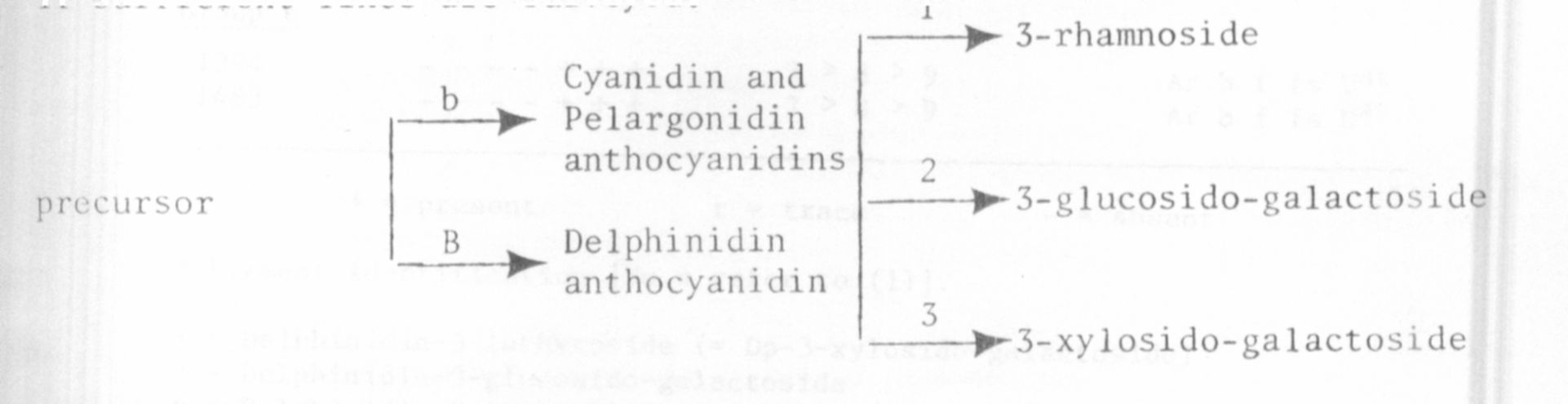
These data suggest that at least three loci are involved in the glyco-
sidation biochemistry of anthocyanins in Pisum seed coats, corresponding
to the steps numbered 1, 2, and 3 in the likely biochemical reaction sequence
shown in the diagram. The data are not inconsistent with the supposition
that Ar controls step 3 (formation of lathyroside). However, with such
meager data the apparent relationship could easily be fortuitous and the
true nature of the genetic control must await the accumulation of breeding
data. On the basis of the present results we might expect to find b plants
with Cyanidin-3-rhamnoside and probably nine groups in both B and b plants
if sufficient lines are surveyed.
(1) Crowden, R. K. 1978. PNL 10:5-8.
(2) Jarman, S. J. and Crowden, R. K. 1973. Phytochemistry 12:171-173.
(3) Harborne, J. B. 1966. Comparative Biochemistry of Flavonoids.
Academic Press, New York.
